Introduction
Aripiprazole is a prescription medication in the atypical antipsychotic class. It was developed in Japan and was approved by the FDA in 2002. The brand name of aripiprazole is Abilify. Aripiprazole is available as a tablet, an orally disintegrating tablet, an oral solution, and a tablet with a sensor (Abilify Mycite). Aripiprazole is also available as a long acting injectable in two formulations called Abilify Maintena and Aristada. There used to be a short acting injectable, but this is no longer available. Even though it falls in the antipsychotic class, it has many uses outside of psychosis.
The FDA approved uses of aripiprazole include the following:
- Schizophrenia
- Acute treatment of manic and mixed episodes of bipolar 1 disorder
- Adjunctive treatment of major depressive disorder
- Irritability associated with autistic disorder
- Tourette’s disorder
Dosing
The dosing of the medication depends on which indication is being treated. Generally, schizophrenia and bipolar mania require higher doses and depression, irritability associated with autism and Tourette’s disorder require lower doses.
| Initial dose | Recommended dose | Maximum dose | |
| Schizophrenia – adults | 10-15 mg | 10-15 mg | 30 mg |
| Schizophrenia – adolescents | 2 mg | 10 mg | 30 mg |
| Bipolar mania – adults, monotherapy | 15 mg | 15 mg | 30 mg |
| Bipolar mania – adults, adjunct to lithium or valproate | 10-15 mg | 15 mg | 30 mg |
| Bipolar mania – pediatric patients as monotherapy or adjunct to lithium or valproate | 2 mg | 10 mg | 30 mg |
| Major depressive disorder – adults, adjunct | 2-5 mg | 5-10 mg | 15 mg |
| Irritability associated with autistic disorder – pediatric patients | 2 mg | 5-10 mg | 15 mg |
| Tourette’s disorder – <50 kg | 2 mg | 5 mg | 10 mg |
| Tourette’s disorder – ≥50 kg | 2 mg | 10 mg | 20 mg |
| Initial dose | Maintenance dose | Dosage forms | Injection site | Dose schedule | Bridge | |
| Abilify Maintena | 400 mg/4 weeks | 300-400mg/4 weeks | Single dose vial or prefilled syringe: 300 and 400mg lyophilized powder | IM deltoid or gluteal, needles provided Non obese Deltoid 1in 23g Glut 1.5in 22g Obese Deltoid 1.5in 22g Glut 2in 21g | 4 weeks | 14 day oral bridge |
| Aristada | 441, 662 or 882mg/4 weeks or 882mg/6 weeks or 1064mg/8 weeks | 441, 662 or 882mg/4 weeks or 882mg/6 weeks or 1064mg/8 weeks | Single-dose prefilled syringe: 441,662,882, or 1064mg Aristado Initio 675mg single-dose prefilled syringe | IM gluteal (441mg can be deltoid) needles provided Deltoid 1in 21g or 1.5in 20g Gluteal 1.5 or 2in 20g | 4,6 or 8 weeks | 21 day oral bridge One 30mg tablet if Initio is used |
Mechanism of action
How aripiprazole works is not completely understood. It is a complicated molecule that binds to many receptors in the brain.
Aripiprazole is a partial dopamine agonist at the dopamine subtype 2 receptor (D2). It also binds to other dopamine subtypes such as D3 and D4. When it comes to schizophrenia, the D2 receptor is often considered the most important dopamine receptor. A partial agonist at D2 receptors means that aripiprazole binds to and activates the receptor to a degree that is less than dopamine, which is the chemical that is supposed to activate the receptor.
This means that dopamine’s effects will be decreased in areas of the brain where dopamine is high like the mesolimbic pathway. This is because although aripiprazole activates dopamine, it does so to a lesser degree than dopamine itself. Decreasing the effects of dopamine here can help with delusions and hallucinations. In areas of the brain where dopamine is low like the mesocortical pathway, aripiprazole will increase the effects of dopamine, because it is a dopamine partial agonist, and partial agonism is better than no agonism. Increasing dopamine here will help with the negative symptoms of schizophrenia.
Although aripiprazole is a partial agonist at D2, it still binds very strongly to the receptor. This means that it can kick off other molecules from the D2 receptor that are weaker binders. To put it in perspective, haloperidol is considered a strong blocker of D2 and has a Ki constant of 1.4 at the D2 receptor. Aripiprazole has a Ki constant of 0.45 at the D2 receptor and binds even stronger than haloperidol (a lower Ki constant means stronger binding).1
Aripiprazole is a 5-HT1A partial agonist. 5-HT1A agonism in clozapine has been shown to increase dopamine levels in the prefrontal cortex. This can be beneficial in the cognitive and negative symptoms of schizophrenia.2
Aripiprazole is a 5-HT2A antagonist. 5-HT2A binding acts as the “brakes” for dopamine release in certain parts of the brain so blocking this receptor also increases dopamine in certain areas of the brain like the striatum.
Aripiprazole is also a 5-HT7 antagonist which may have antidepressant and pro-cognitive effects.
Aripiprazole has little affinity at histamine receptors, which often cause sedation in other antipsychotic medications. Aripiprazole has no affinity for M1 muscarinic receptors and will not cause anticholinergic side effects.
Side effects
Black box warnings
Antipsychotics as a class have a warning against their use in elderly patients with dementia-related psychosis. The increased risk of death is from cerebrovascular adverse reactions such as stroke.

Increased suicidality in children and young adults is a black box warning found on all antidepressant medications. The increased risk was seen in patients age 24 and younger and is always important to assess for in patients with mental illness.
Warnings and precautions
- Neuroleptic malignant syndrome: Antipsychotics can rarely cause a potentially fatal side effect called neuroleptic malignant syndrome (NMS). NMS consists of high temperature, muscle rigidity, mental status changes and autonomic instability (such as changes in pulse and blood pressure). Incidence of NMS is as low as 0.02% for antipsychotics as a class and case reports do exist with aripiprazole as the causative agent.4
- Tardive dyskinesia: Antipsychotics can cause a movement disorder called tardive dyskinesia (TD). TD is characterized by involuntary movements especially seen in the mouth and tongue. The risk of TD is three times lower in second generation antipsychotics compared to first generation antipsychotics.5 Aripiprazole is not commonly associated with TD, but there are still case reports of this side effect presenting after aripiprazole use.
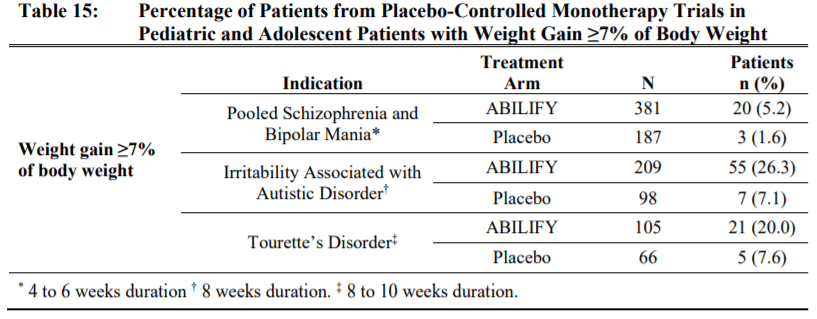
- Metabolic changes: Antipsychotics as a class can cause metabolic changes such as high blood sugar, high cholesterol and body weight changes. Compared to many other second generation antipsychotics, aripiprazole has a lower risk to cause these changes. Hyperglycemia and dyslipidemia is mostly comparable to placebo, so lets focus on weight gain. In adults, there is a chance of weight gain for example in patients with schizophrenia, 8.1% of aripiprazole treated patients gained 7% body weight compared to 3.2% of placebo treated patients. Pediatric patients had a higher chance of gaining weight. Weight gain was seen in 20% or more being treated for Tourette’s or irritability associated with Autism.
- Pathologic gambling and other compulsive behaviors: Post-marketing case reports showed that aripiprazole may be associated with compulsive behaviors such as gambling, sexual activity, or shopping. Due to the mechanism, dopamine can increase in some pathways in the brain which can lead to compulsive behaviors. This is not a side effect seen with most other antipsychotics, but brexpiprazole (Rexulti) also has this warning.
- Orthostatic hypotension: Aripiprazole can cause blood pressure changes and dizziness. Orthostatic hypotension was seen in 1% of aripiprazole treated patients and 0.3% of placebo treated patients.
- Falls: Changes in blood pressure and dizziness can lead to falls.
- Leukopenia, neutropenia, and agranulocytosis: Lowering of white blood cells and other cell counts can be seen in antipsychotic use. It is common to have blood drawn for a complete blood count (CBC) at least annually while on antipsychotics.
- Seizures: Antipsychotics can lower the seizure threshold. 0.1% of patients treated with aripiprazole had seizures.
- Potential for cognitive and motor impairment: Aripiprazole can be sedating in some people. Somnolence was seen in 11% of aripiprazole treated patients compared to 6% of placebo treated patients. Patients should not drive or operate machinery until they know how aripiprazole affects them.
Side effects
The most common adverse reactions in adult patients in clinical trials (≥10%) were nausea,
vomiting, constipation, headache, dizziness, akathisia, anxiety, insomnia, and restlessness.
The most common adverse reactions in the pediatric clinical trials (≥10%) were somnolence,
headache, vomiting, extrapyramidal disorder, fatigue, increased appetite, insomnia, nausea,
nasopharyngitis, and weight gain.
Evidence per indication
Schizophrenia
Aripiprazole compared to a first generation antipsychotic
First, we will look at an old study that was published in 2002. This study included 414 patients with a diagnosis of schizophrenia or schizoaffective disorder who had failed an antipsychotic medication other than clozapine. Patients were randomly assigned to one of four groups: aripiprazole 15 mg, aripiprazole 30 mg, haloperidol 10 mg, or placebo. Treatments were given after breakfast for four weeks. The PANSS score was used to track symptoms.
The PANSS decreases per group were as follows:
- Placebo: -2.9
- Aripiprazole 15 mg: -15.5
- Aripiprazole 30 mg: -11.4
- Haloperidol 10 mg: -13.8
The side effects that had the biggest difference from placebo were headache, anxiety, insomnia, nausea, dizziness, vomiting, and somnolence.6
Is this 11-15 point difference clinically significant? The minimum difference in PANSS score to be clinically significant is estimated to be 15 points.7
Aripiprazole compared to another second generation antipsychotic
We saw aripiprazole compared to a first generation antipsychotic. How will it fare compared to a second generation antipsychotic?
The next study included 404 patients who were randomized to receive aripiprazole 20 mg, aripiprazole 30 mg, risperidone 6 mg or placebo for four weeks. Participants in this study had to have had a previous response to an antipsychotic in the past other than clozapine.
The PANSS decreases per group were as follows:
- Placebo: -5.0
- Aripiprazole 20 mg: -14.5
- Aripiprazole 30 mg: -13.9
- Risperidone 6 mg: -15.7
17% in the placebo group discontinued treatment due to side effects, 8% in the risperidone group, 11% in the aripiprazole 20-mg group, and 8% in the aripiprazole 30-mg group.
Side effects were common in all four groups: 91% (92/101) in the aripiprazole 20-mg group, 91% (91/100) in the aripiprazole 30-mg group, 93% (92/99) in the risperidone group, and 86% (89/103) in the placebo group.
The side effects that had the biggest difference from placebo were headache, vomiting, and akathisia.8
What doses are effective?
So we can see that aripiprazole has comparable efficacy to both first generation and second generation antipsychotics. Next, I wanted to see if lower doses could be effective.
The next study compared different doses of aripiprazole in the treatment of an acute exacerbation of schizophrenia. The patients were given either aripiprazole 2, 5, or 10 mg daily or placebo for 6 weeks.
The PANSS decreases per group were as follows:
- Placebo: -5.3
- Aripiprazole 2 mg: -8.2
- Aripiprazole 5 mg: -10.6
- Aripiprazole 10 mg: -11.3
This study shows that the higher doses are more effective in treating acute exacerbations of schizophrenia, and this is shown in the recommended dosing in the package insert.9
How effective is it long-term?
The next study is a 52-week extension to a 26-week, multicenter, randomized trial in patients with chronic schizophrenia. The patients were either on aripiprazole (15-30 mg per day) or olanzapine (10-20 mg/day).
The PANSS decreases per group were as follows in stable patients:
- Aripiprazole: -7.94
- Olanzapine: -7.36
The PANSS decreases per group were as follows in unstable patients:
- Aripiprazole: -31.19
- Olanzapine: -29.55
Aripiprazole has less extrapyramidal symptoms (10% vs 18% on olanzapine). Olanzapine caused more weight gain than aripiprazole.
Aripiprazole was effective for the long-term control of schizophrenia with less EPS and weight gain than olanzapine.10
Can aripiprazole be added to other antipsychotics?
Using more than one antipsychotic in a patient is controversial. Most of the guidelines do not recommend it outside of adding another antipsychotic to clozapine in very refractory cases. Though in practice, you will commonly see providers use more than one antipsychotic. Let’s see what the evidence says.
First, I wanted to look at the least controversially treatment – adding aripiprazole to clozapine.
The first study took place in Seoul, Korea with only 62 patients. The patients were on clozapine treatment for at least 1 year, were on a stable dose for at least 8 weeks and were taking at least 400 mg of clozapine daily unless they had side effects. Patients were then randomly assigned to 8 weeks of aripiprazole between 5-30 mg/day or placebo. The brief psychiatric rating scale was used to assess efficacy.11
BPRS decrease per group:
- clozapine/aripiprazole: -5.1
- clozapine/placebo: -4.7
Another study did find a benefit of adding aripiprazole to patients already taking clozapine. This was an even smaller study with only 27 participants. Patients were on clozapine 100-900 mg/day for at least 12 months and were assigned to receive aripiprazole 15 mg/day for 16 weeks. The total PANSS decreased from 74.7 to 60.3 at the end of the 16 weeks (-14.4).12
This conflicting evidence makes it difficult to know which patients would benefit from the addition of aripiprazole to clozapine.
The next studies will look at adding aripiprazole to other second generation antipsychotics.
This study added aripiprazole (2-15 mg) or placebo to patients with an inadequate response to risperidone (4-8 mg) or quetiapine (400-800 mg). The PANSS decreased by 8.8 in the aripiprazole group and 8.9 in the placebo group.13
Finally, it is thought that aripiprazole may prevent the weight gain caused by other antipsychotics.
A small study of 16 patients stable on olanzapine for at least 1 month added aripiprazole 15 mg/day or placebo to see if weight would be effected. The median length of treatment with olanzapine was 71 months and the mean olanzapine dose was 22 mg daily. This is a high dose for a long period of time. The placebo group’s weight increased by 2.1 lbs while the aripiprazole group had a decrease in weight of 2.9 lbs. 14
What are the differences in the two long acting injectable options?
Abilify Maintena and Aristada are two aripiprazole products that are approved for use in patients with schizophrenia
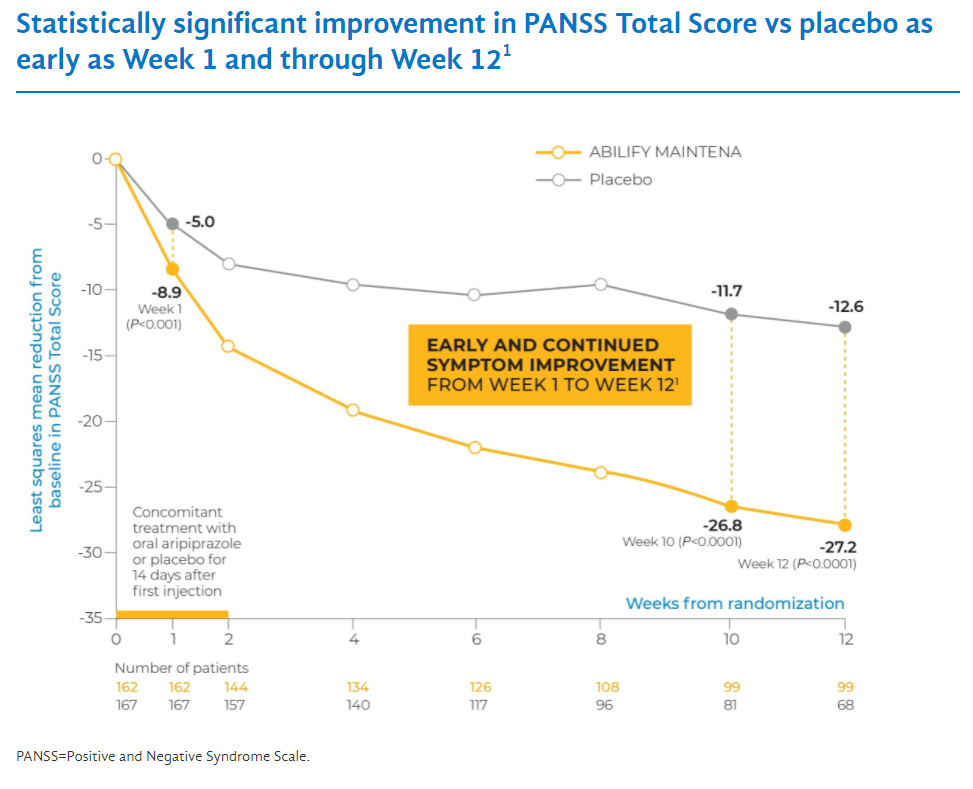
Let’s compare how effective each one is. In the Abilify Maintena trial, patients had an initial PANSS score of 103. 12 weeks into treatment the average PANSS score decreased by 27.2 points. In the Aristada trial, the baseline PANSS score was around 92. 12 weeks into treatment the PANSS score decreased by around 21 points. So it is not comparing apples to apples because the patients were more sick in the Abilify Maintena trial. Abilify Maintena has some advantages such as also being approved for bipolar disorder, and Aristada has some advantages such as the option to dose every two months rather than every month.
Bipolar disorder
Mania Monotherapy
First, let’s look at if aripiprazole is effective by itself in controlling bipolar mania.
A study compared aripiprazole 15-30 mg/day to placebo, or lithium 900-1500 mg/day. The Young Mania Rating Scale (YMRS) was used to assess efficacy and a 6.6 point decrease in the YMRS is the goal for a change to be clinically significant.15 A YMRS difference was seen between the placebo and aripiprazole at day two. The aripiprazole score decreased by 4.3 while the placebo decreased by 2.8. At week three the aripiprazole group decreased by 12.6, the lithium group by 12.0 and the placebo group by 9.0. At week 12, 56.5% of the aripiprazole group had responded to the medication and 49% had responded to the lithium.
Over the 12-week study, side effects higher than 10% in the aripiprazole group were as follows: headache (23.4%), nausea (22.7%), akathisia (14.9%), sedation (13.0%), and constipation (10.4%).16
A Cochrane review combined 10 studies with 3340 patients to get a bigger picture view of aripiprazole’s efficacy in treating bipolar mania. A high dropout rate of >20% may have effected the estimate of efficacy, but the dropout rate was around the same in the placebo groups. Aripiprazole beat placebo by 3.66 points on the YMRS at 3 weeks, but no statistically significant difference was seen at 6 weeks.
Compared with placebo, aripiprazole caused more movement disorders, gastrointestinal disturbances and constipation. Additionally it caused more children/adolescents to have a prolactin level that fell below the lower limit of normal.17
Aripiprazole added to lithium or depakote
Another option with antipsychotic medication in bipolar disorder is to add them to a mood stabilizer. This study looked at adding aripiprazole to lithium or depakote.
Patients were on either lithium or depakote and this study added either aripiprazole or placebo to their regimen. Relapse rates at 52 weeks were 17% in the aripiprazole group and 29% in the placebo group. This study shows that continuing aripiprazole, prevents people from having a relapse of symptoms. The most common side effects in the aripiprazole group compared to the placebo group were: headache (13.2% versus 10.8%), weight increase (9.0% versus 6.6%), tremor (6.0% versus 2.4%), and insomnia (5.4% versus 9.6%).18
Bipolar depression
People with bipolar disorder spend more of their time in a depressed state than a manic state so it is very important to have effective treatments for bipolar depression. Let’s see how aripiprazole faired.
The first study was 2 identically designed, 8-week, randomized, double-blind placebo controlled trials. A depression scale called the Montgomery-Asberg Rating Scale was used to compare the efficacy. Aripiprazole was dosed between 5-30 mg/day and at week 8 there was not a statistically significant difference compared to placebo. Not only that but the dropout rate was very high. In study 1 the dropout rate for aripiprazole vs placebo was 46.8% vs 35.1% and in study 2 it was 41.2% (aripiprazole) vs 29.8% placebo.19
Another study confirmed aripiprazole’s efficacy in bipolar mania but showed aripiprazole was not superior to placebo for preventing depressive episodes at 26 and 100 weeks of maintenance therapy.20
Depression
Although aripiprazole does not seem to be effective in treating bipolar depression, it does seem effective in treating major depressive disorder as an add-on medication.
Aripiprazole adjunct vs placebo in treating depression
The first study looked at patients on SSRI or SNRI antidepressants who had an inadequate response and were still symptomatic. These patients then had either aripiprazole 2-20 mg or placebo added to their regimen. The MADRS total score decreased in the aripiprazole group by 10.1 and by 6.4 in the placebo group. The study concluded that adjunctive aripiprazole was associated with a two-fold higher remission rate than adjunctive placebo and is an effective add-on medication for depression.21
So now that we know aripiprazole beats placebo as as add-on medication, lets see how it does compared to another antidepressant.
Aripiprazole adjunct vs antidepressant adjunct in treating depression
The next study compared adding aripiprazole 2.5-20 mg/day (average dose 2.99 mg) vs bupropion 150-300 mg/day to patients who where unresponsive to SSRI monotherapy. Participants had at least moderately severe depressive symptoms after 4 weeks or more of SSRI treatment. At week 6, the remission rate was 55.4% in the aripiprazole group compared to 34.0% in the bupropion group. This study shows that aripiprazole may be more effective than other add-on therapies.22
Aripiprazole adjunct vs switching to another antidepressant
Generally, if someone has a partial response to an antidepressant, the options are to add on another medication or switch and try something completely new. Is adding aripiprazole more effective than switching regimens? A study looked at this.
First, we will review a prospective, randomized, rater-blinded, flexible-dose, 6-week, multi-center study. The efficacy of adding aripiprazole 2-15 mg vs switching antidepressants was compared in patients who were previously adequately treated but who had an inadequate response to their current antidepressants. The MADRS score decreased by 16.3 in the aripiprazole group and by 7.6 in the switch group showing a significant difference in favor of adding aripiprazole.23
Aripiprazole in treatment resistant depression
Sometimes medications don’t work for people. Once a person fails a medication when they are taking it for an adequate duration at an adequate dose, they are considered treatment resistant. A small retrospective chart review of 30 patients who had failed an antidepressant, and one atypical antipsychotic showed that 14/30 patients were either much improved or very much improved once starting aripiprazole. A 46.7% response rate doesn’t seem high, but it may still be a useful tool in treatment resistant depression.24
Suicidality
Suicidality is a difficult symptom to treat. In fact, antidepressants (including aripiprazole) have a black box warning for increased risk of suicidal behavior and thoughts in those 24 years old or younger. That being said, a post hoc analysis demonstrated that adjunctive aripiprazole treatment for depression in patients with an inadequate response to antidepressant medication is associated with a decreased rate of suicidality in a group of subjects not at significant risk. So aripiprazole may have slight suicidality benefits in those 25 years old or older.25
Irritability associated with autistic disorder
There are only two medications that are FDA approved to treat behaviors associated with autism and aripiprazole is one of them.
Aripiprazole vs placebo
218 children and adolescents age 6-17 with a diagnosis of autism were randomized to receive either aripiprazole 5 mg, 10 mg, 15 mg, or placebo. The participants had behaviors such as tantrums, aggression, and/or self injurious behaviors.
The Aberrant Behavior Checklist Irritability subscale was used to assess changes in behavior over the 8-week trial. The decrease of scores can be seen below.
- Aripiprazole 5 mg: -12.4
- Aripiprazole 10 mg: -13.2
- Aripiprazole 15 mg: -14.4
- Placebo: -8.4
Aripiprazole was effective and generally well tolerated, with sedation being the most common reason to stop it in this patient population.26
Aripiprazole vs risperidone
59 children were randomized to receive either risperidone or aripiprazole for 2 months. The Aberrant Behavior Checklist (ABC) was used to assess changes in behavior. The mean dose of aripiprazole was 5.5 mg/day and risperidone was 1.12 mg/day. The safety and efficacy was comparable between the two medications and both were found to be effective.27
Long term efficacy
The first study had two phases. In phase 1, aripiprazole was dosed between 2-15 mg for 13-26 weeks. If the patients responded (25% decrease in Aberrant Behavior Checklist) they could move on to phase two. In phase 2, the patients were either continued on aripiprazole or switched to placebo and continued until relapse of irritability symptoms or up to 16 weeks. Relapse rates at week 16 were 35% for aripiprazole and 52% for placebo but the difference was not statistically significant.28
Next, there was a 52-week open label flexible dose (2-15 mg/day) study. New participants in the study had an 8.0 point decrease in their Aberrant Behavior Checklist scores, while continuing patients maintained there score decreases for the entire 52 weeks. 29
What behaviors actually improve?
A study broke down the individual measures in the Aberrant Behavior Checklist to see which specific symptoms were improved. It found the following symptoms had statistically significant improvements.
- Mood changes quickly
- Cries/screams inappropriately
- Stamps feet/bangs objects30
Tourette’s disorder
Only a few antipsychotics are FDA approved to treat Tourette’s disorder. Haloperidol and pimozide are the first-generation approved options and aripiprazole stands alone as the only approved second generation option.
Aripiprazole vs placebo
A study recruited patients aged 7-17 from 76 sites in four countries. Patients were randomized to three groups: low-dose aripiprazole (5 mg/day if <50 kg; 10 mg/day if ≥50 kg), high-dose aripiprazole (10 mg/day if <50 kg; 20 mg/day if ≥50 kg), or placebo for 8 weeks. The primary endpoint was to analyze the mean change from baseline to week 8 in the Yale Global Tic Severity Scale Total Tic Score (YGTSS-TTS).
Least-squared mean changes were found to be statistically significant compared to placebo (high dose -9.9, low dose -6.3. This translated to 69% (29/42) of patients in the low-dose and 74% (26/35) of patients in the high-dose aripiprazole groups being scored as very much improved or much improved compared with 38% (16/42) in the placebo group.
The most common adverse events were sedation (low dose 18.2%, high dose 8.9%, placebo 2.3%), somnolence (low dose 11.4%, high dose 15.6%, placebo 2.3%), and fatigue (low dose 6.8%, high dose 15.6%, placebo, 0%).31
References
- Goodnick, P. J., & Jerry, J. M. (2002). Aripiprazole: profile on efficacy and safety. Expert Opinion on Pharmacotherapy, 3(12), 1773–1781.
- Tuplin E, Holahan M. Aripiprazole, A Drug that Displays Partial Agonism and Functional Selectivity. Curr Neuropharmacol. 2017 Nov; 15(8): 1192–1207.
- Kales HC, Valenstein M, Kim HM, McCarthy JF, Ganoczy D, Cunningham F, Blow FC. Mortality risk in patients with dementia treated with antipsychotics versus other psychiatric medications. Am J Psychiatry. 2007 Oct;164(10):1568-76; quiz 1623. doi: 10.1176/appi.ajp.2007.06101710. PMID: 17898349.
- Agrawal A, Bajaj D, Bajaj S, Mattana J. Aripiprazole Induced Late Neuroleptic Malignant Syndrome. Am J Ther. 2019 Nov/Dec;26(6):e772-e773. doi: 10.1097/MJT.0000000000000944. PMID: 30730330.
- O’Brien A. Comparing the risk of tardive dyskinesia in older adults with first-generation and second-generation antipsychotics: a systematic review and meta-analysis. Int J Geriatr Psychiatry. 2016 Jul;31(7):683-93. doi: 10.1002/gps.4399. Epub 2015 Dec 17. PMID: 26679687.
- Kane J, Carson W, Saha A, et al. Efficacy and Safety of Aripiprazole and Haloperidol Versus Placebo in Patients With Schizophrenia and Schizoaffective Disorder. J Clin Psychiatry. 2002 Sep;63(9):763-71.
- Hermes E, Sokoloff D, Stroup T, et al. Minimum Clinically Important Difference In The Positive And Negative Syndrome Scale Using Data From The CATIE Schizophrenia Trial. J Clin Psychiatry. 2012 Apr; 73(4): 526–532.
- Potkin SG, Saha AR, Kujawa MJ, Carson WH, Ali M, Stock E, Stringfellow J, Ingenito G, Marder SR. Aripiprazole, an antipsychotic with a novel mechanism of action, and risperidone vs placebo in patients with schizophrenia and schizoaffective disorder. Arch Gen Psychiatry. 2003 Jul;60(7):681-90.
- Cutler AJ, Marcus RN, Hardy SA, O’Donnell A, Carson WH, McQuade RD. The efficacy and safety of lower doses of aripiprazole for the treatment of patients with acute exacerbation of schizophrenia. CNS Spectr. 2006 Sep;11(9):691-702; quiz 719.
- Chrzanowski WK, Marcus RN, Torbeyns A, Nyilas M, McQuade RD. Effectiveness of long-term aripiprazole therapy in patients with acutely relapsing or chronic, stable schizophrenia: a 52-week, open-label comparison with olanzapine. Psychopharmacology (Berl). 2006 Dec;189(2):259-66.
- Chang JS, Ahn YM, Park HJ, Lee KY, Kim SH, Kang UG, Kim YS. Aripiprazole augmentation in clozapine-treated patients with refractory schizophrenia: an 8-week, randomized, double-blind, placebo-controlled trial. J Clin Psychiatry. 2008 May;69(5):720-31.
- Mitsonis CI, Dimopoulos NP, Mitropoulos PA, Kararizou EG, Katsa AN, Tsakiris FE, Katsanou MN. Aripiprazole augmentation in the management of residual symptoms in clozapine-treated outpatients with chronic schizophrenia: An open-label pilot study. Prog Neuropsychopharmacol Biol Psychiatry. 2007 Mar 30;31(2):373-7.
- Kane JM, Correll CU, Goff DC, Kirkpatrick B, Marder SR, Vester-Blokland E, Sun W, Carson WH, Pikalov A, Assunção-Talbott S. A multicenter, randomized, double-blind, placebo-controlled, 16-week study of adjunctive aripiprazole for schizophrenia or schizoaffective disorder inadequately treated with quetiapine or risperidone monotherapy. J Clin Psychiatry. 2009 Oct;70(10):1348-57.
- Henderson DC, Fan X, Copeland PM, Sharma B, Borba CP, Boxill R, Freudenreich O, Cather C, Evins AE, Goff DC. Aripiprazole added to overweight and obese olanzapine-treated schizophrenia patients. J Clin Psychopharmacol. 2009 Apr;29(2):165-9.
- Lukasiewicz M, Gerard S, Besnard A, Falissard B, Perrin E, Sapin H, Tohen M, Reed C, Azorin JM; Emblem Study Group. Young Mania Rating Scale: how to interpret the numbers? Determination of a severity threshold and of the minimal clinically significant difference in the EMBLEM cohort. Int J Methods Psychiatr Res. 2013 Mar;22(1):46-58.
- Keck PE, Orsulak PJ, Cutler AJ, Sanchez R, Torbeyns A, Marcus RN, McQuade RD, Carson WH; CN138-135 Study Group. Aripiprazole monotherapy in the treatment of acute bipolar I mania: a randomized, double-blind, placebo- and lithium-controlled study. J Affect Disord. 2009 Jan;112(1-3):36-49.
- Brown R, Taylor MJ, Geddes J. Aripiprazole alone or in combination for acute mania. Cochrane Database Syst Rev. 2013 Dec 17;(12):CD005000.
- Marcus R, Khan A, Rollin L, Morris B, Timko K, Carson W, Sanchez R. Efficacy of aripiprazole adjunctive to lithium or valproate in the long-term treatment of patients with bipolar I disorder with an inadequate response to lithium or valproate monotherapy: a multicenter, double-blind, randomized study. Bipolar Disord. 2011 Mar;13(2):133-44.
- Thase ME, Jonas A, Khan A, Bowden CL, Wu X, McQuade RD, Carson WH, Marcus RN, Owen R. Aripiprazole monotherapy in nonpsychotic bipolar I depression: results of 2 randomized, placebo-controlled studies. J Clin Psychopharmacol. 2008 Feb;28(1):13-20.
- Yatham LN. A clinical review of aripiprazole in bipolar depression and maintenance therapy of bipolar disorder. J Affect Disord. 2011 Jan;128 Suppl 1:S21-8.
- Berman RM, Fava M, Thase ME, Trivedi MH, Swanink R, McQuade RD, Carson WH, Adson D, Taylor L, Hazel J, Marcus RN. Aripiprazole augmentation in major depressive disorder: a double-blind, placebo-controlled study in patients with inadequate response to antidepressants. CNS Spectr. 2009 Apr;14(4):197-206.
- Cheon EJ, Lee KH, Park YW, Lee JH, Koo BH, Lee SJ, Sung HM. Comparison of the Efficacy and Safety of Aripiprazole Versus Bupropion Augmentation in Patients With Major Depressive Disorder Unresponsive to Selective Serotonin Reuptake Inhibitors: A Randomized, Prospective, Open-Label Study. J Clin Psychopharmacol. 2017 Apr;37(2):193-199.
- Han C, Wang SM, Kwak KP, Won WY, Lee H, Chang CM, Tang TC, Pae CU. Aripiprazole augmentation versus antidepressant switching for patients with major depressive disorder: A 6-week, randomized, rater-blinded, prospective study. J Psychiatr Res. 2015 Jul-Aug;66-67:84-94.
- Barbee JG, Conrad EJ, Jamhour NJ. Aripiprazole augmentation in treatment-resistant depression. Ann Clin Psychiatry. 2004 Oct-Dec;16(4):189-94.
- Weisler RH, Khan A, Trivedi MH, Yang H, Eudicone JM, Pikalov A, Tran QV, Berman RM, Carlson BX. Analysis of suicidality in pooled data from 2 double-blind, placebo-controlled aripiprazole adjunctive therapy trials in major depressive disorder. J Clin Psychiatry. 2011 Apr;72(4):548-55.
- Marcus RN, Owen R, Kamen L, Manos G, McQuade RD, Carson WH, Aman MG. A placebo-controlled, fixed-dose study of aripiprazole in children and adolescents with irritability associated with autistic disorder. J Am Acad Child Adolesc Psychiatry. 2009 Nov;48(11):1110-1119.
- Ghanizadeh A, Sahraeizadeh A, Berk M. A head-to-head comparison of aripiprazole and risperidone for safety and treating autistic disorders, a randomized double blind clinical trial. Child Psychiatry Hum Dev. 2014;45(2):185-92.
- Findling RL, Mankoski R, Timko K, Lears K, McCartney T, McQuade RD, Eudicone JM, Amatniek J, Marcus RN, Sheehan JJ. A randomized controlled trial investigating the safety and efficacy of aripiprazole in the long-term maintenance treatment of pediatric patients with irritability associated with autistic disorder. J Clin Psychiatry. 2014 Jan;75(1):22-30.
- Marcus RN, Owen R, Manos G, Mankoski R, Kamen L, McQuade RD, Carson WH, Corey-Lisle PK, Aman MG. Aripiprazole in the treatment of irritability in pediatric patients (aged 6-17 years) with autistic disorder: results from a 52-week, open-label study. J Child Adolesc Psychopharmacol. 2011 Jun;21(3):229-36.
- Aman MG, Kasper W, Manos G, Mathew S, Marcus R, Owen R, Mankoski R. Line-item analysis of the Aberrant Behavior Checklist: results from two studies of aripiprazole in the treatment of irritability associated with autistic disorder. J Child Adolesc Psychopharmacol. 2010 Oct;20(5):415-22.
- Sallee F, Kohegyi E, Zhao J, McQuade R, Cox K, Sanchez R, van Beek A, Nyilas M, Carson W, Kurlan R. Randomized, Double-Blind, Placebo-Controlled Trial Demonstrates the Efficacy and Safety of Oral Aripiprazole for the Treatment of Tourette’s Disorder in Children and Adolescents. J Child Adolesc Psychopharmacol. 2017 Nov;27(9):771-781.